We know you want what’s best for the patients you treat. You want options that provide the best outcomes with the fewest side effects and require the least amount of time and discomfort for your patient. And of course, you are looking out for their pocketbooks as well.
Alpha-Stim neuromodulation devices are proven to be safe and effective for treating anxiety, insomnia, depression, and acute, chronic, and post-traumatic pain.
A Simpler Way to Relieve Anxiety, Insomnia, Depression & Pain
Alpha-Stim devices are non-drug, non-invasive, and use cranial electrotherapy stimulation (CES) and Microcurrent Electrical Therapy (MET) to help treat anxiety, insomnia, depression, and pain, safely and effectively.
Alpha-Stim AID, a CES device, is ideal for patients with anxiety, insomnia, and depression, while Alpha-Stim M is uniquely designed to provide both CES and MET modalities and is able to treat anxiety, insomnia, depression, and pain. Prescribed by a healthcare provider, Alpha-Stim safely provides rapid, effective, and lasting symptom relief without the risks of dangerous side effects or addiction. The device is designed to be used anywhere – at home, in a provider’s office, or any location.
Clinically Proven Results with Alpha-Stim

Improved Sleep Quality
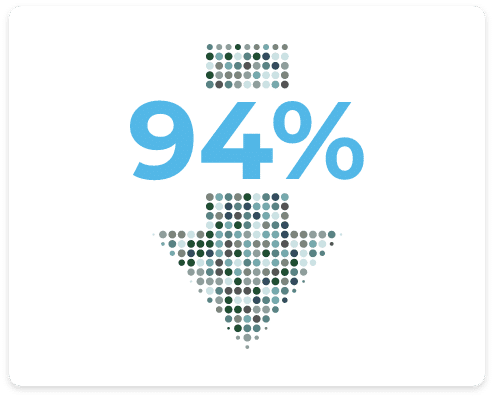
Overall Decrease in Anxiety
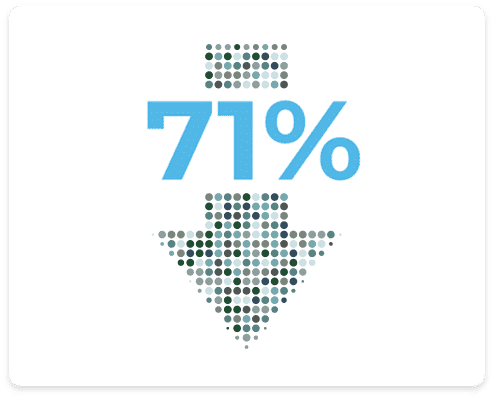
Significant Pain Reduction
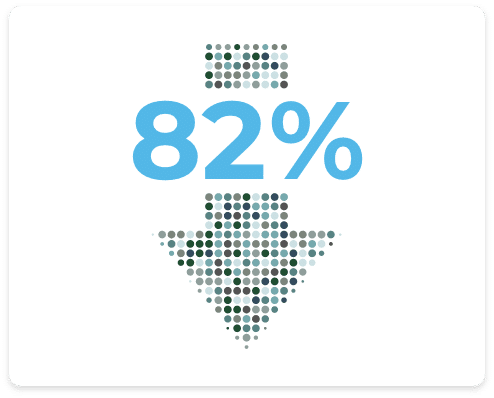
Decrease in Depression
Healthcare Practitioners
Try Alpha-Stim today. Talk to one of our reps to get started.
For practitioners new to Alpha-Stim, we have options for trying both Alpha-Stim AID and Alpha-Stim M. Typically, an in-office or clinic trial can help you identify those individuals who readily respond to Alpha-Stim treatment, and if Alpha-Stim is a good fit for your practice.
Use the form below to get more information about Alpha-Stim and connect with a local Alpha-Stim representative.
How Alpha-Stim Works
Alpha-Stim uses two types of stimulation
Cranial Electrotherapy Stimulation (CES) for anxiety, insomnia, and depression, and Microcurrent Electrical Therapy (MET) for pain.
Cranial electrotherapy stimulation (CES) is a form of non-invasive brain stimulation that treats psychiatric disorders such as anxiety and insomnia. Alpha-Stim CES applies a patented waveform using rhythmic, low-amplitude current to the head via earclip electrodes.
Psychiatric disorders, such as anxiety, are associated with alterations in electric activity in large-scale brain networks. Research suggests that Alpha-Stim CES modulates large-scale brain network activity patterns, like those seen in anxiety, in two synergistic ways:

Ultimately, these two ways of stimulating the brain normalize the pathological signaling in the default mode network observed in many psychiatric disorders, providing rapid, effective, and lasting symptom relief without the side effects of medication.
Patients using Alpha-Stim for pain use Smart Probes or apply self-adhesive electrodes around the part of the body where the pain originates from to deliver MET.
Explore Alpha-Stim for Anxiety, Insomnia, Depression & Pain
Alpha-Stim® M
Acute, chronic and post-traumatic pain, and anxiety, insomnia, and depression.
Alpha-Stim® AID
Designed to to help patients suffering from anxiety, insomnia, and depression.
What Patients & Providers Say
About Alpha-Stim
Former President, American Association of Police Officers; Former President, Homeland Crisis InstituteTexas





A More Holistic Approach to Mental Health
“I’m starting to understand then that there’s more of a sort of 360 holistic approach to anxiety, so psychological and also behaviorally, environment, that type of thing. And it became more of like, yes, talking therapy is really important to understand what’s going on. But if we just talked about the anxiety, then it’s like, not much changes…It’s then like, well, there’s more to this as well. What does technology offer? And that’s where I found Alpha-Stim. And I’ve been using Alpha-Stim within the practice probably for, must be at least seven years now.”
Moving from talk therapy to a more holistic approach that includes Alpha-Stim, Mike Ward of the London & Hampshire Anxiety Clinics shares why he shifted how he approaches anxiety treatment to include more – talking therapies, technology, neuroscience, brain, body, and a 360-degree health approach.
-
1 Lichtbroun AS, Raicer MMC, Smith RB. The treatment of fibromyalgia with Cranial Electrotherapy Stimulation. Journal of Clinical Rheumatology. 2001; 7(2):72-78.
-
2,4 Barclay TH, Barclay RD. A clinical Trial of cranial electrotherapy stimulation for anxiety and comorbid depression. Journal of Affective Disorders. 2014;164:171-177. Presented at the American Psychological Association National Conference, Honolulu, July 2013.
-
3 Holubec JT. Cumulative response from Cranial Electrotherapy Stimulation (CES) for chronic pain. Practical Pain Management. 2009; 9(9):80-83.


